 At the first energy level, the only orbital available to electrons is the 1s orbital, but at the second level, as well as a 2s orbital, there are also orbitals called 2p orbitals.Ī p orbital is rather like 2 identical balloons tied together at the nucleus. Not all electrons inhabit s orbitals (in fact, very few electrons live in s orbitals). The nearer the nucleus the electrons get, the lower their energy.ģs, 4s (etc) orbitals get progressively further from the nucleus. The effect of this is to slightly reduce the energy of electrons in s orbitals. ("Electron density" is another way of talking about how likely you are to find an electron at a particular place.)Ģs (and 3s, 4s, etc) electrons spend some of their time closer to the nucleus than you might expect. If you look carefully, you will notice that there is another region of slightly higher electron density (where the dots are thicker) nearer the nucleus. This is similar to a 1s orbital except that the region where there is the greatest chance of finding the electron is further from the nucleus - this is an orbital at the second energy level. s orbitals are spherically symmetric around the nucleus - in each case, like a hollow ball made of rather chunky material with the nucleus at its centre. The "s" tells you about the shape of the orbital. The "1" represents the fact that the orbital is in the energy level closest to the nucleus. 
The orbital occupied by the hydrogen electron is called a 1s orbital. What is the electron doing in the orbital? We don't know, we can't know, and so we just ignore the problem! All you can say is that if an electron is in a particular orbital it will have a particular definable energy. 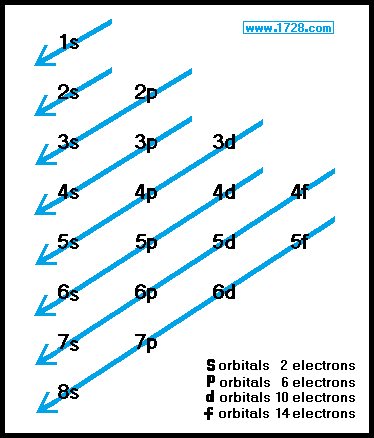
Note: If you wanted to be absolutely 100% sure of where the electron is, you would have to draw an orbital the size of the Universe! You can think of an orbital as being the region of space in which the electron lives. Such a region of space is called an orbital. The diagram shows a cross-section through this spherical space.ĩ5% of the time (or any other percentage you choose), the electron will be found within a fairly easily defined region of space quite close to the nucleus. In the hydrogen case, the electron can be found anywhere within a spherical space surrounding the nucleus. You keep on doing this over and over again, and gradually build up a sort of 3D map of the places that the electron is likely to be found. You have no idea how it got from the first place to the second. Soon afterwards, you do the same thing, and find that it is in a new position. Suppose you had a single hydrogen atom and at a particular instant plotted the position of the one electron. Note: In this diagram (and the orbital diagrams that follow), the nucleus is shown very much larger than it really is. If something is impossible, you have to accept it and find a way around it. That makes it impossible to plot an orbit for an electron around a nucleus. The Heisenberg Uncertainty Principle (not required at A'level) says - loosely - that you can't know with certainty both where an electron is and where it's going next. Equally it's no use knowing that it is travelling at 500 mph due west if you have no idea whether it is near Iceland or the Azores at that particular moment. Note: In order to plot a plane's course, it is no use knowing its exact location in mid-Atlantic if you don't know its direction or speed. To plot a path for something you need to know exactly where the object is and be able to work out exactly where it's going to be an instant later. 
The impossibility of drawing orbits for electrons It is essential that you understand the difference between them. Orbits and orbitals sound similar, but they have quite different meanings. You could straighten the circles out and draw the electronic structure as a simple energy diagram. The circles show energy levels - representing increasing distances from the nucleus. As you will see in a moment, it is impossible to know exactly how they are actually moving. It gives the impression that the electrons are circling the nucleus in orbits like planets around the sun. It is, however, a simplification and can be misleading. Note: There are many places where you could still make use of this model of the atom at A' level. In any introductory chemistry course you will have come across the electronic structures of hydrogen and carbon drawn as: 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
